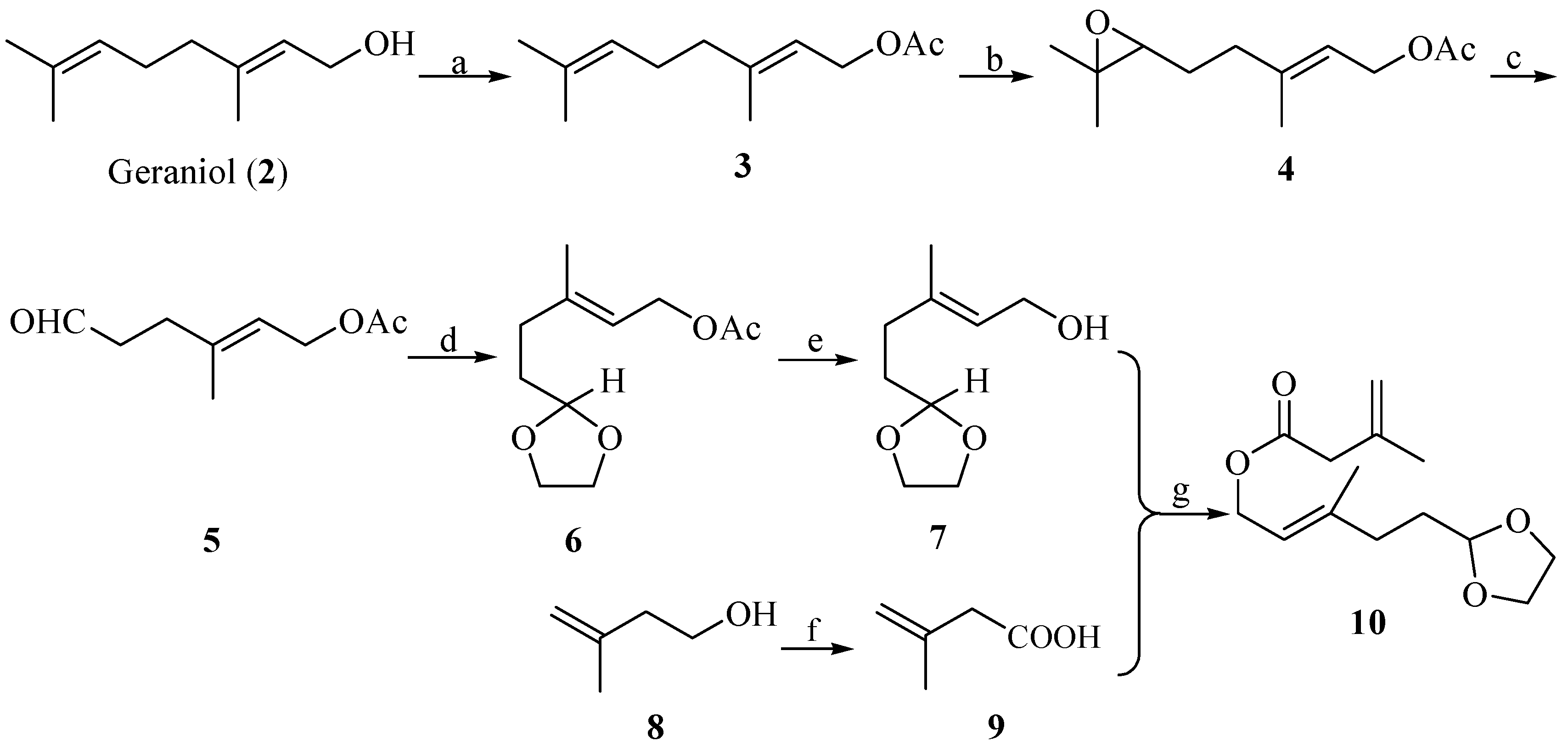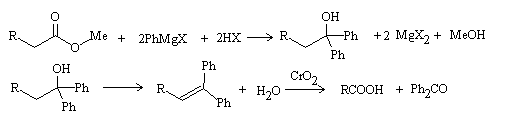
Steroids are lipids derived from the triterpenoid lanosterol Structures are based on a tetracyclic ring system Four rings designated A, B, C, and D Numbering. - ppt download
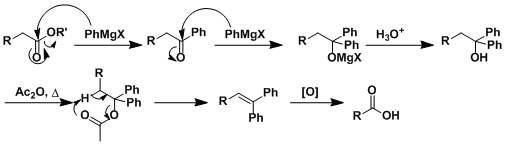
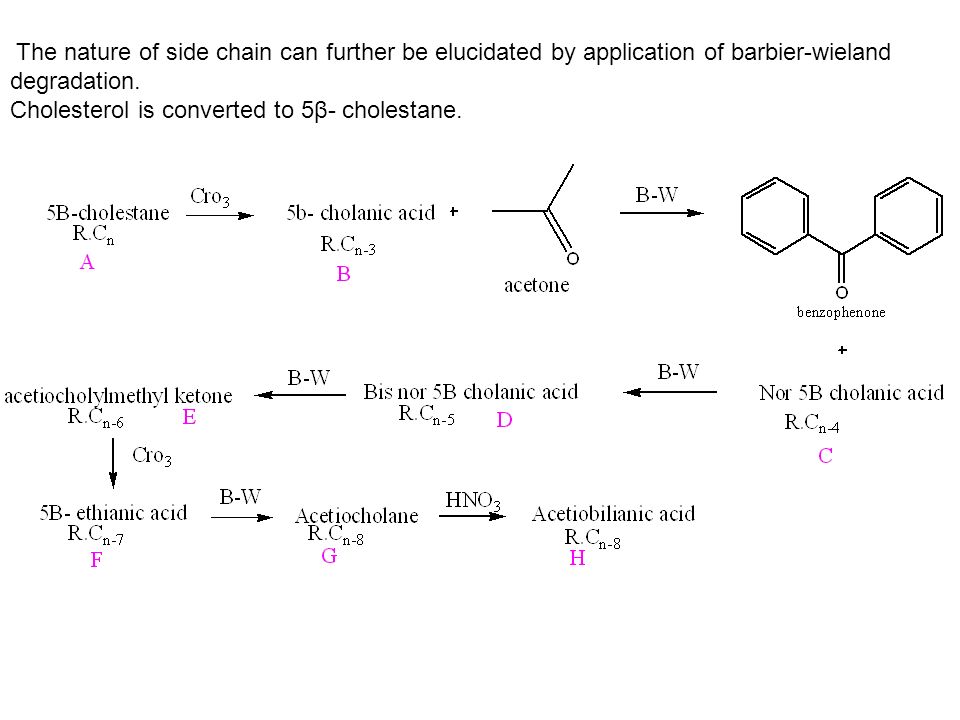
STUDY OF BARBIER-WIELAND DEGRADATION AND ARNDT-EISTERT SYNTHESIS OF LIGNOCERIC ACID* BY T. R. KASTURI AND B. H. E'ER SUMMARY
STUDY OF BARBIER-WIELAND DEGRADATION AND ARNDT-EISTERT SYNTHESIS OF LIGNOCERIC ACID* BY T. R. KASTURI AND B. H. E'ER SUMMARY
Steroids Derived from Bile Acids. XX. Degradation of 3α,9α-epoxy-11-Ketonorcholanic Acid to 3α,9α-Epoxy-11-ketoetiocholanic
![PDF] Terpenoid biosynthesis from 1-deoxy-D-xylulose in higher plants by intramolecular skeletal rearrangement. | Semantic Scholar PDF] Terpenoid biosynthesis from 1-deoxy-D-xylulose in higher plants by intramolecular skeletal rearrangement. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/65842b3095cbddc16efdcd9d52013d0e8d21d50c/5-Figure7-1.png)
PDF] Terpenoid biosynthesis from 1-deoxy-D-xylulose in higher plants by intramolecular skeletal rearrangement. | Semantic Scholar