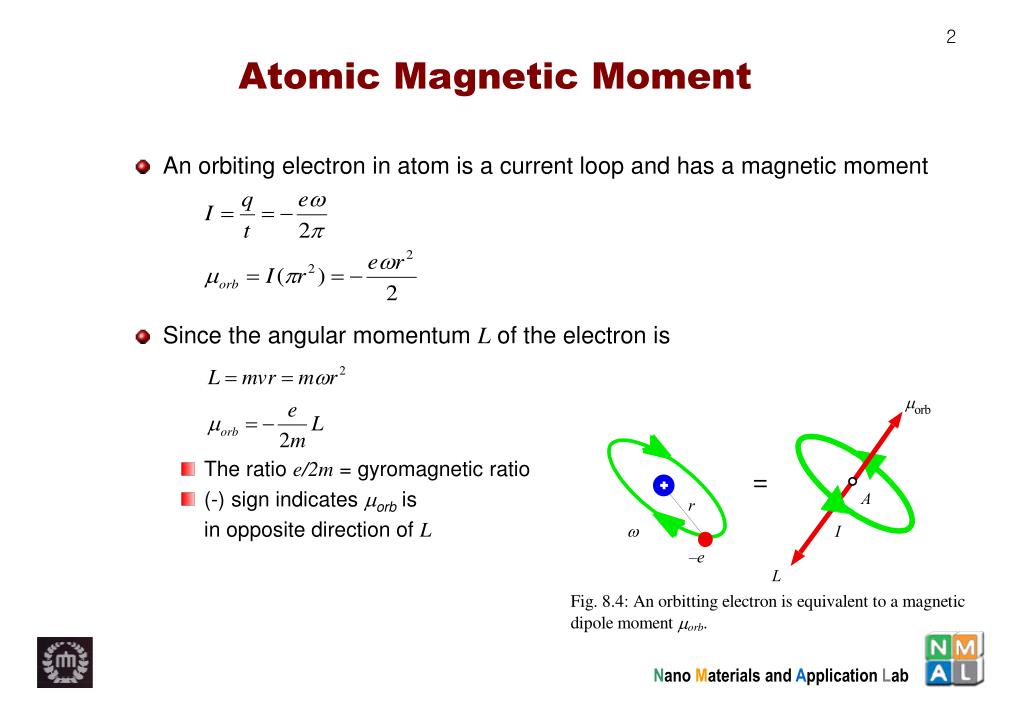
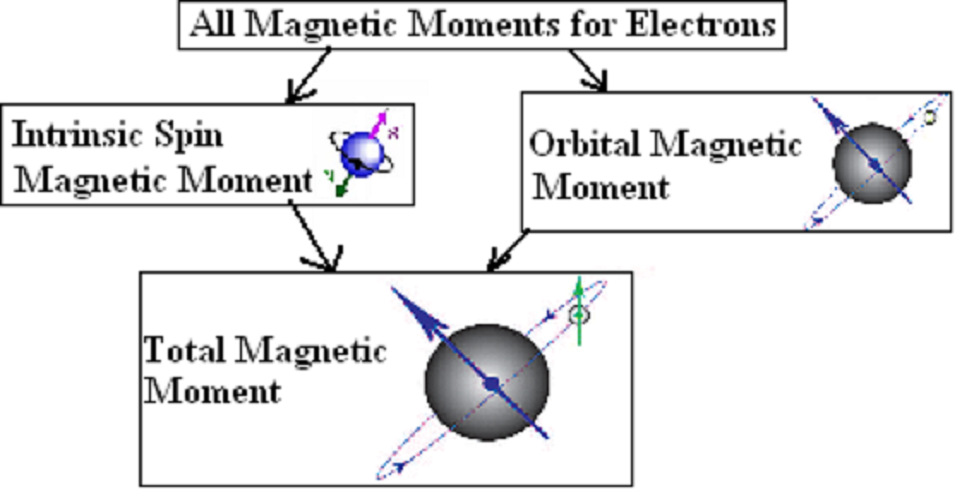
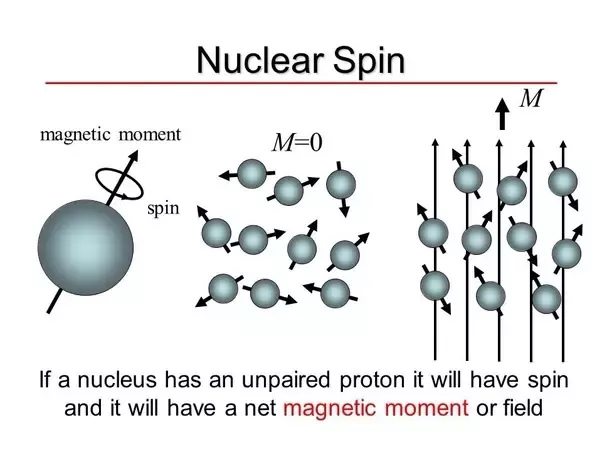
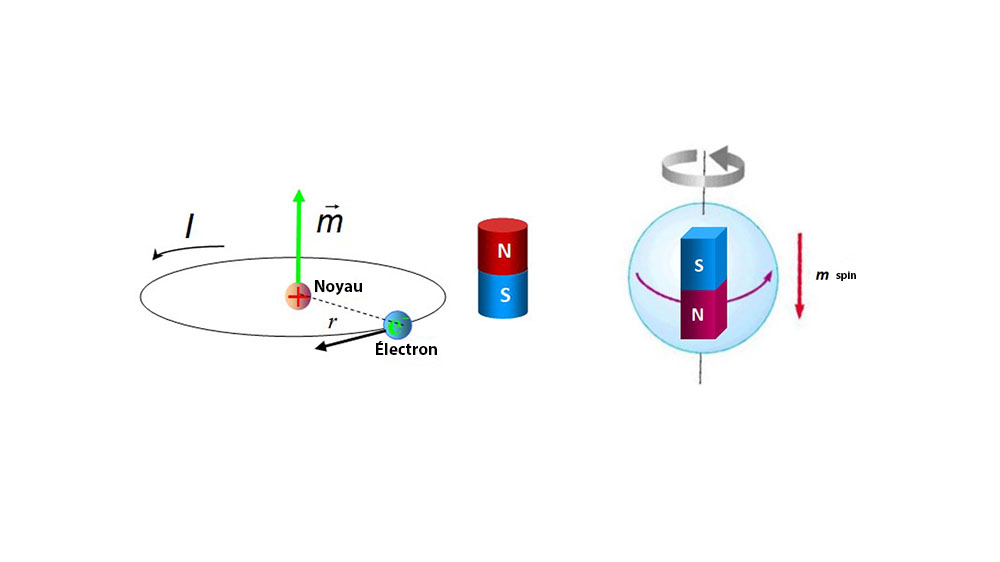
1: Magnetic moment on the atomic scale. (A) In a free atom the magnetic... | Download Scientific Diagram

quantum mechanics - Ignoring spin, what is its orbital magnetic moment of an electron in a hydrogen atom in the 2p orbital? - Physics Stack Exchange

Physics - Ch 66.5 Quantum Mechanics: The Hydrogen Atom (34 of 78) Magnetic Moment in Hydrogen - YouTube

Assume that each iron atom has a permanent magnetic moment equal to 2 Bohr magnets (1 Bohr mag ≠ nequals 9.27 × 10^−24 Am^2). The density of atoms in iron is 8.52 ×