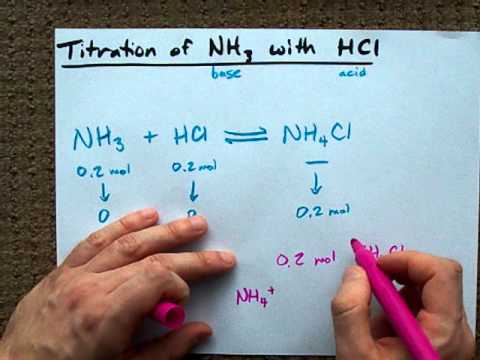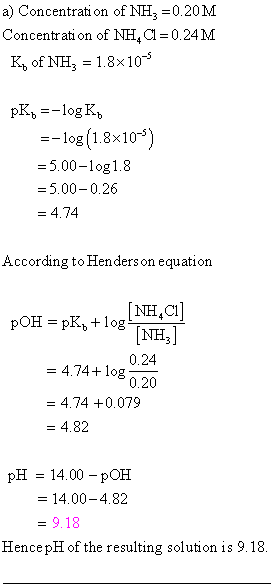
OneClass: A)Calculate the pH of the 0.20 M NH3/0.24 M NH4Cl buffer. B)What is the pH of the buffer af...

OneClass: does the addition of HCl to a NH4Cl/ NH3 buffer solution produce the common ion effect? why...

OneClass: Consider a buffer solution that is 0.50 M in NH3 and 0.20 M in NH4Cl. For ammonia, pKb=4.75...

Table 1 from VOLTAMMETRIC BEHAVIOURS OF Zn(II) AND Ni(II) COMPLEXES WITH ACID RED 1 AT MERCURY ELECTRODE | Semantic Scholar

PPT – Calculate the pH of the 0.30 M NH3/0.36 M NH4Cl buffer system. What is the pH after the addition of 20.0 mL of 0.050 M NaOH to 80.0 mL of




![Solved: BUFFERS 10. An Ammonium[NH3]/ammonium Chloride (NH... | Chegg.com Solved: BUFFERS 10. An Ammonium[NH3]/ammonium Chloride (NH... | Chegg.com](https://media.cheggcdn.com/media/98f/s907x655/98fc93a3-c6f6-4a89-8acb-45d7d9f5eb4d/image.png)